|
This means that the nucleus of a beryllium atom will pull on the outermost electrons with more force, effectively compressing the distance between itself and these electrons. However, beryllium has #4# protons in its nucleus, as opposed to #3# protons which are present in the nucleus of a lithium atom. The same is true for the two outermost electrons in beryllium, they are screened by two core electrons. In lithium's case, the outermost electron is being screened from the nucleus by two core electrons. 
The effective nuclear charge is simply a measure of the net positive charge that affects the outermost electrons in an atom. That happens because beryllium has a higher effective nuclear charge, #Z_"eff"#, than lithium. which ultimately determine the atomic radius, are closer to the nucleus in beryllium's case. Thank you.Lithium, #"Li"#, and beryllium, #"Be"#, are both located in period 2 of the periodic table, in group 1 and group 2, respectively.įor both these elements, the outermost electrons are located on the second energy level in the 2s-subshell. This is for iodine, and the last configuration that we have is five p. Next we have the configuration as five as to 40 10 5 p. The charges symbol capital in if you talk of the second part here, the outermost configuration is three s. So in the first part we are given with the ultimate configuration as three S. The item actually as for the atomic configuration given. Now let's move on to the second part of the question in which we need to identify the elements. Next we have the element and it would be leased for potassium, then calcio, joe, mania, arsenic, selenium blooming and krypton. Then it will be for flooring and maximum will be for neon which is a noble gas as we have arranged it according to this period trend. In the first part we have like this lithium will be having at least ionic radius, then it will be for beryllium, then boron, then carbon, nitrogen, oxygen. 
Atomic shell for electron increases accordingly. Whereas if you talk in groups, when we go down a group on going down, going down in a group of periodic tables, the ionic radius off the items increases as atomic shell. The ionic radius of the elements decreases each room. So what happens is in going across a period. In this question we have to define the trend for ionic radius when we grow across a period on up theater when we move across it. 5s'4d'Sp5 5s24d105p' 3523p' which Is Iocated in the block Element Q would be which Is located In the block Element would which Is located in the ~block Element would which Is located in the ~block Element X would would be expected to have the highest_ first Ionlzation energy and Of these elements, to have the lowest first ionization energy? would be expected element 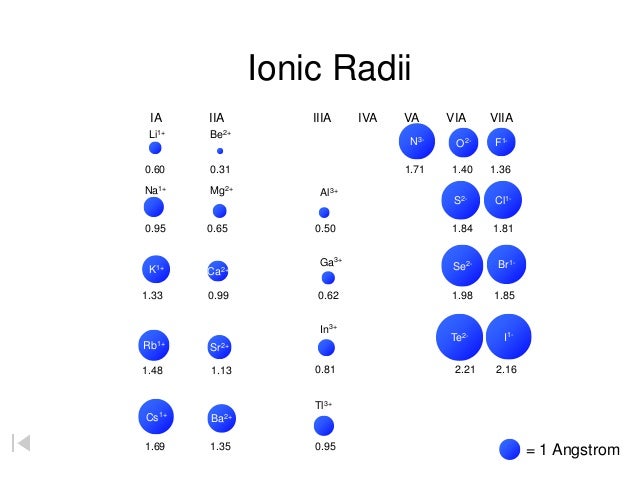
Ignoring the Kp4 smallest atomic radii (ignore noble gases) In what group number Is it located? What is the name of this group? main-group elements: Q, R, T and Xwlth the outer electron 12, Consider the four hypothetical Then answer the questions that follow: configurations indicated below. 
Arrange each of the following groups of elements from smallest-to-largest atomic radii N, B, Li, Be C, F,0 K, As, Kr Ca, Se, Br, Ge, Ga Br, Ge Se, Cs, Sr, Sb As, Bi N, P Ca, Sr, Rb, Br, Cs Ne In, Sn Se, Sr, Cs, Sb, elements in Period which element on the Periodic Table should have the 11.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
